After years in development, BGU Negev scientists have developed a breakthrough nanochip, which could propel cancer immunotherapy forward.
Their findings were published Friday in the prestigious peer-reviewed journal Science Advances.
This study has two innovative and important aspects – one from a nanotechnology perspective and the other from a biological perspective.

"The field of nanotechnology took off about 20 years ago, primarily stemming from a need to reduce the size of components on computer processing chips," says principal investigator Prof. Mark Schvartzman (pictured above). "Nowadays, the field offers unique tools that serve scientists from many different fields. These tools allow us to create, view and control objects just 10 nanometers or less in size. That is the size-scale of a single biomolecule in the body. We achieved unprecedented control setting up a molecule as a single cell and we even managed to "suppress" or "activate" important processes in the cell for the first time."
The study also has important ramifications for medicine and biology.
"We managed to understand how the size and physical arrangement of the receptors on the cell affect how the white blood cells "talk to" the other cells in the body," explains co-author Prof. Angel Porgador, dean of the Faculty of Health Sciences at BGU. "This understanding is of far-reaching significance in developing immunotherapy treatments against cancer. Today, cell activities within the body can be directed to fight cancer by genetically engineering receptors. This innovative approach is at the forefront of cancer treatments and has been proven effective for certain types of blood cancers, but there is a need to develop other methods that are effective against different types of cancer. That development requires a deep understanding of how the immune system operates. Today, that understanding comes about through new nanotechnology tools."

Above left to right, Post-doc student Dr. Guillaume Le Saux, doctoral student Esti Toledo, Prof. Mark Schvartzman and Prof. Angel Porgador
Lymphocytes, a type of white blood cell, are the gatekeepers of our immune system. Like guards on patrol who stop suspicious people on occasion, lymphocytes glide through the blood in the body and know how to differentiate between healthy cells and sick cells, which could turn cancerous. If the cell is identified as sick – infected by a virus or cancerous – 'the patrol' breaks them up and destroys them. If the cell signals that it is healthy, the white blood cells continue as usual.
To differentiate sick cells from healthy cells, the white blood cells are equipped with receptors – molecules which know how to connect to other molecules on the suspicious cells (ligands). There are receptors that when connecting to a ligand signal the white blood cell to attack the suspicious cell, and there are receptors called immune checkpoints that signal to suppress an attack and "save" the cell. The delicate balance between signals enables the immune system to differentiate between cells and prevent healthy cells from being attacked. Discovery of the immune attack suppressors has led in recent years to the immunotherapy approach to tackling cancer, which helps the body identify and attack cancer cells. This approach blocks and neutralizes the immune checkpoints and enables the lymphocytes to attack the cancer cells masquerading as healthy cells.
Despite the vast amount of research in the field, there are still several open questions concerning the activities of these immune checkpoint inhibitors, such as: Does the distance between the receptor that signals attack and the receptor that signals do not attack affect the attack suppression? If so, how? A breakthrough study by BGU scientists sheds light on these questions. They developed a silicone microchip whose surface mimics an artificial cancer cell, which can attach the receptors and control their organization.
Prof. Schvartzman, a member of the Department of Materials Engineering and the Ilse Katz Institute for Nanoscale Science & Technology at BGU, developed the unique chip in his lab. Using nanolithography, whose origins lie in creating computer processors, the researchers created a chip with metal etchings just 10 nanometers in size. The researchers then glued both types of ligands to the chip. The spacing of the ligands was varied in a controlled manner and to ensure that only one molecule attached to each receptor.
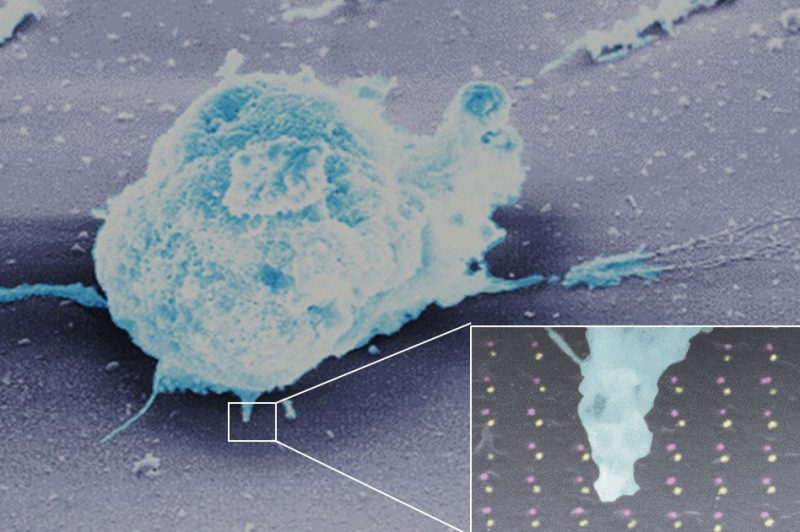
Above: The chip (Photo Credit: Esti Toledo and Dr. Guillaume Le Saux)
The team was able to confirm that lymphocyte cells interacted with the chip as if it were a cancerous cell and attempted to destroy it. They discovered that the distance between receptors had a dramatic effect on the lymphocytes' response: the farther away the receptors for attack and suppression, the greater the suppression. The researchers were greatly surprised by this finding because it contradicts the prevailing understanding of the scientific community, that proximity is required to suppress an attack. According to the researchers, the cell membrane has limited flexibility and therefore when the receptors are in proximity, the lymphocyte cannot attach to both receptors.
The research was carried out by doctoral student Esti Toledo and post-doc student Dr. Guillaume Le Saux with the assistance of research groups from Germany and France.
The research was funded by the Israel Science Foundation (Grant nos. 1401/15 and 2058/18), the Ministry of Science and Technology, and the Israel Cancer Association.
Media Coverage:
The Times of Israel
JPost
Israel 355 News